Aqueous Sodium Chloride Reacts With Aqueous Lead Ii Nitrate
When these two solutions are mixed the lead II cations. The precipitate lead chloride is insoluble in cold water but it is soluble in hot water.
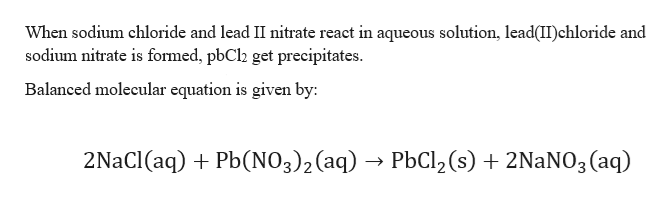
Answered When Sodium Chloride And Lead Ii Bartleby
Elements Atoms And Ions 4 Nomenclature 5 Measurements And Calculations 6 Chemical Composition 7 Chemical Reactions.
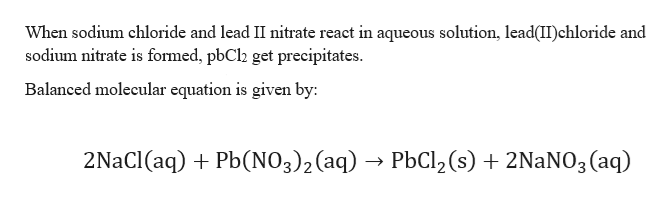
. When aqueous solutions of sodium sulfate and lead II nitrate are mixed lead sulfate precipitates out of soultion. You would expect one of the products of this reaction to be. PbNO 3 2 aq 2 NaCl aq 2 NaNO 3 aq PbCl 2 s What is the molarity of the sodium chloride solution if 880 mL of it will produce 695 g of the leadII chloride.
Aqueous sodium chloride reacts with aqueous lead nitrate to yield a lead chloride precipitate and aqueous sodium nitrate are. Sodium sulfate ironII chloride calcium perchlorate barium hydroxide lead nitrate. Sodium sulfide Na 2 S is added to leadII nitrate PbNO 3 2.
2NH4Cl aq PbNO32 aq ---- 2NH4NO3 aq PbCl2 s When aqueous copper II chloride reacts with aqueous ammonium phosphate copper II phosphate precipitates out of solution Given the net. The reactants are. Strontium Chloride with Sodium Sulfide.
Aqueous solutions of leadII nitrate and sodium chloride react to form solid leadII chloride and aqueous sodium nitrate according to the reaction below. The reaction equation for this chemical reaction is as follows. For reaction 2 we have Cu 2 Cl Na and SO 4 2 in solution.
II Chloride with Sodium Sulfide Play Movie duration 85 seconds. In this case lead II nitrate PbNO32 and sodium iodide NaI both soluble in water will exist as ions in aqueous solution. 1 A double displacement reaction takes place when aqueous sodium carbonate reacts with aqueous tin II nitrate.
Aqueous zinc chloride plus aqueous sodium hydroxide produce aqueous sodium chloride and solid zinc hydroxide. The chloride makes a precipitate with the Pb². Be sure to include the states of each of the reactants and products.
2NaClaq ZnOH2s Refer to the related link below. Lead II chloride PbCl₂ s Sodium nitrate NaNO₃ aq Salts form nitrate are soluble. The reaction equation for this chemical reaction is as follows.
ZnCl2aq 2NaOHaq --. Click to see full answer. When an aqueous solution of leadII nitrate reacts with a solution of sodium chloride leadII chloride precipitate and aqueous sodium nitrate are formed.
This is the type of reaction called double displacement reaction because ions from which reactants are made go through a double exchange to produce new chemical compounds. If the precipitate contains lead write the balanced chemical equation for this reaction. The result is a thick black precipitate.
Most chlorides and sulphates are soluble according to the table. The chemical equation for. Aqueous barium nitrate reacts with sulfuric acid H 2 SO 4.
An Introduction 2 Matter 3 Chemical Foundations. 2 Na aq CO 3 2 aq Cu 2 aq 2 Cl aq CuCO 3 s 2 Na aq 2 Cl aq Note that sodium chloride does not precipitate and we write it as ions in the equation. Brooks Cole Cengage Learning.
Youre dealing with a double replacement reaction in which two soluble ionic compounds in aqueous solution react to form an insoluble solid that precipitates out of solution. Aqueous barium chloride and aqueous potassium carbonate react to produce solid barium carbonate and aqueous potassium chloride. When sodium chloride solution is added to lead nitrate solution then it results in the formation of a precipitate of lead chloride and sodium nitrate.
Aqueous lead II nitrate Pb NO32 undergoes a double displacement reaction with aqueous sodium chloride NaCl in which a precipitate forms. SnCO32 2 For the reaction 3 Cu 8HNO3 --. An Introduction 8.
Aqueous sodium chloride reacts with aqueous lead II nitrate to yield a lead II chloride precipitate and aqueous sodium nitrate sodium chloride lead II nitrate lead II chloride sodium nitrate NaCl PbNO 3 2 PbCl 2 NaNO 3 2 NaCl aq PbNO 3 2 aq PbCl 2 ppt 2 NaNO 3 aq 2. By kinetic particle theory you will end up with a solid of lead ii chloride which is insoluble in an aqueous solution of iron ii. When sodium chloride solution is added to lead nitrate solution then it results in the formation of a precipitate of lead chloride and sodium nitrate.
BaCl2aq K2co3 - Ba co3s 2 KClaq Aqueous sodium oxalate and aqueous leadii nitrate react to produce solid leadii oxalate and aqueous sodium nitrate. Lead II nitrate Pb NO₃₂ aq Sodium chloride NaCl aq The products are. Write and balance the chemical equation.
Likewise does a reaction occur when aqueous solutions of Chromium II. A precipitate is expected when an aqueous solution of potassium iodide is added to an aqueous solution of. The precipitate lead chloride is insoluble in cold water but it is soluble in hot water.
Ba NO32 aq H2SO4 - BaSO4 downward arrow 2HNO3 aq Hope this help. Combining aqueous solutions of lead ii nitrate and iron ii chloride Im assuming you mean iron ii chloride because its simpler to mess around with will result in an aqueous solution of lead ii iron ii nitrate and chloride ions. The reaction is happening between copper II chloride and lead II nitrate in an aqueous solution.
Silver Nitrate with Sodium Sulfide Play Movie duration 10 seconds.

Solved Lead Ii Nitrate Reacts With Sodium Chloride In Aqueous Solution T0 Form Precipitate What Is The Net Ionic Equation For This Reaction Na Aq No Aq Nano S Pb T Aq 2ci Aq Pbclz S Na Aq

How To Write The Net Ionic Equation For Pb No3 2 Nacl Youtube


Comments
Post a Comment